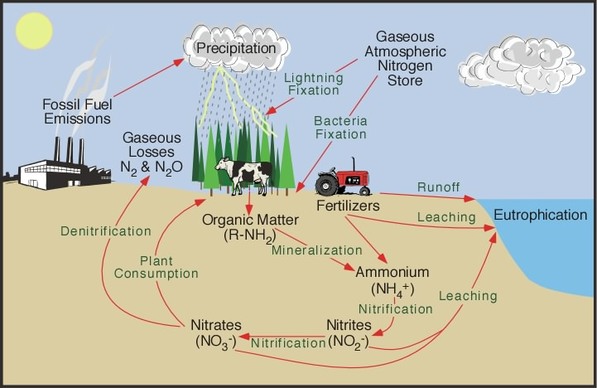
The store of nitrogen found in the atmosphere, where it exists as a gas (mainly N2), plays an important role for life. This store is about one million times larger than the total nitrogen contained in living organisms. Other major stores of nitrogen include organic matter in soil and the oceans. Despite its abundance in the atmosphere, nitrogen is often the most limiting nutrient for plant growth.
In most ecosystems nitrogen is primarily stored in living and dead organic matter. This organic nitrogen is converted into inorganic forms when it re-enters the biogeochemical cycle via decomposition. Decomposers, found in the upper soil layer, chemically modify the nitrogen found inorganic matter from ammonia (NH3 ) to ammonium salts (NH4+ ). This process is known as mineralization and it is carried out by a variety of bacteria, actinomycetes, and fungi. Nitrogen in the form of ammonium can be absorbed onto the surfaces of clay particles in the soil. When released, most of the ammonium is often chemically altered by a specific type of autotrophic bacteria into nitrite (NO2- ). Further modification by another type of bacteria converts the nitrite to nitrate (NO3- ). Both of these processes involve chemical oxidation and are known as nitrification. However, nitrate is very soluble and it is easily lost from the soil system by leaching. Some of this leached nitrate flows through the hydrologic system until it reaches the oceans where it can be returned to the atmosphere by denitrification. Denitrification is also common in anaerobic soils and is carried out by heterotrophic bacteria. The process of denitrification involves the metabolic reduction of nitrate into nitrogen (N2) or nitrous oxide (N2O) gas. Both of these gases then diffuse into the atmosphere. Almost all of the nitrogen found in any terrestrial ecosystem originally came from the atmosphere. Significant amounts enter the soil in rainfall or through the effects of lightning. The majority is biochemically fixed within the soil by specialized microorganisms like bacteria,actinomycetes, and cyanobacteria. Members of the bean family and some other kinds of plants form mutualistic symbiotic relationships with nitrogen fixing bacteria. In exchange for some nitrogen, the bacteria receive from the plants carbohydrates and special structures in roots where they can exist in a moist environment. Scientists estimate that biological fixation globally adds approximately 140 million metric tons of nitrogen to ecosystems every year.
In most ecosystems nitrogen is primarily stored in living and dead organic matter. This organic nitrogen is converted into inorganic forms when it re-enters the biogeochemical cycle via decomposition. Decomposers, found in the upper soil layer, chemically modify the nitrogen found inorganic matter from ammonia (NH3 ) to ammonium salts (NH4+ ). This process is known as mineralization and it is carried out by a variety of bacteria, actinomycetes, and fungi. Nitrogen in the form of ammonium can be absorbed onto the surfaces of clay particles in the soil. When released, most of the ammonium is often chemically altered by a specific type of autotrophic bacteria into nitrite (NO2- ). Further modification by another type of bacteria converts the nitrite to nitrate (NO3- ). Both of these processes involve chemical oxidation and are known as nitrification. However, nitrate is very soluble and it is easily lost from the soil system by leaching. Some of this leached nitrate flows through the hydrologic system until it reaches the oceans where it can be returned to the atmosphere by denitrification. Denitrification is also common in anaerobic soils and is carried out by heterotrophic bacteria. The process of denitrification involves the metabolic reduction of nitrate into nitrogen (N2) or nitrous oxide (N2O) gas. Both of these gases then diffuse into the atmosphere. Almost all of the nitrogen found in any terrestrial ecosystem originally came from the atmosphere. Significant amounts enter the soil in rainfall or through the effects of lightning. The majority is biochemically fixed within the soil by specialized microorganisms like bacteria,actinomycetes, and cyanobacteria. Members of the bean family and some other kinds of plants form mutualistic symbiotic relationships with nitrogen fixing bacteria. In exchange for some nitrogen, the bacteria receive from the plants carbohydrates and special structures in roots where they can exist in a moist environment. Scientists estimate that biological fixation globally adds approximately 140 million metric tons of nitrogen to ecosystems every year.